Alex Constantine - May 20, 2010
“ ... 'Over the last five years the field has seen a 100-fold increase in the length of genetic material wholly constructed from raw chemicals,' said synthetic biologist Drew Endy of Stanford University. 'This is over six doublings in the max length of a genome that can be constructed.' ... "
by Rachel Swaby | Wired | May 20, 2010
 Man-made DNA has booted up a cell for the first time.
Man-made DNA has booted up a cell for the first time.
In a feat that is the culmination of two and a half years of tests and adjustments, researchers at the J. Craig Venter Institute inserted artificial genetic material — chemically printed, synthesized and assembled — into cells that were then able to grow naturally.
“We all had a very good feeling that it was going to work this time,” said Venter Institute synthetic biologist Daniel Gibson, co-author of the study published May 20 in Science. “But we were cautiously optimistic because we had so many letdowns following the previous experiments.”
On a Friday in March, scientists inserted over 1 million base pairs of synthetic DNA into Mycoplasma capricolum cells before leaving for the weekend. When they returned on Monday, their cells had bloomed into colonies.
“When we look at life forms, we see fixed entities,” said J. Craig Venter, president of the Institute, in a recent podcast. “But this shows in fact how dynamic they are. They change from second to second. And that life is basically the result of an information process. Our genetic code is our software.”
Coaxing the software to power a cell proved harder than expected.

After the Venter Institute announced in early 2008 that it had assembled a synthetic Mycoplasma genitalium genome, the assumption was that it would be running cells in no time. But this particular cell type, despite its minimal size, was not an ideal research partner. One problem was speed.
“We had to deal with the fact that M. genitalium had an extremely slow growth rate,” Gibson said. “For every experiment that was done, it took more than a month to get results.”
Moreover, transplanting the code into recipient cells was failing. So researchers cut their losses and called in a substitute, opting for the larger, speedier and less finicky Mycoplasma mycoides. The choice was a good one.
“Over the last five years the field has seen a 100-fold increase in the length of genetic material wholly constructed from raw chemicals,” said synthetic biologist Drew Endy of Stanford University. “This is over six doublings in the max length of a genome that can be constructed.”
Plunging costs of synthesis allowed a leap past the 1 million base-pair mark, from code to assembly. “Imagine doubling the diameter of a silicon wafer that can be manufactured that much, going from 1 cm to 1 meter [fabrications] in just five years,” Endy said. “That would have been an incredible achievement.”
“They rebuilt a natural sequence and they put in some poetry,” said University of California at San Francisco synthetic biologist Chris Voigt. “They recreated some quotes in the genome sequence as watermarks.”
It’s an impressive trick, no doubt, but replicating a natural genome with a little panache is also the limit of our present design capabilities.
Researchers, for instance, figure yeast can handle the assembly of 2 million base pairs, but they’re not sure about more. And an energy-producing cyanobacteria that sequesters carbon, Gibson says, is still several years off.
The ultimate goal, of course, is a brand-new genome from the ground up. Now, Voigt said, “what do you do with all that design capacity?”
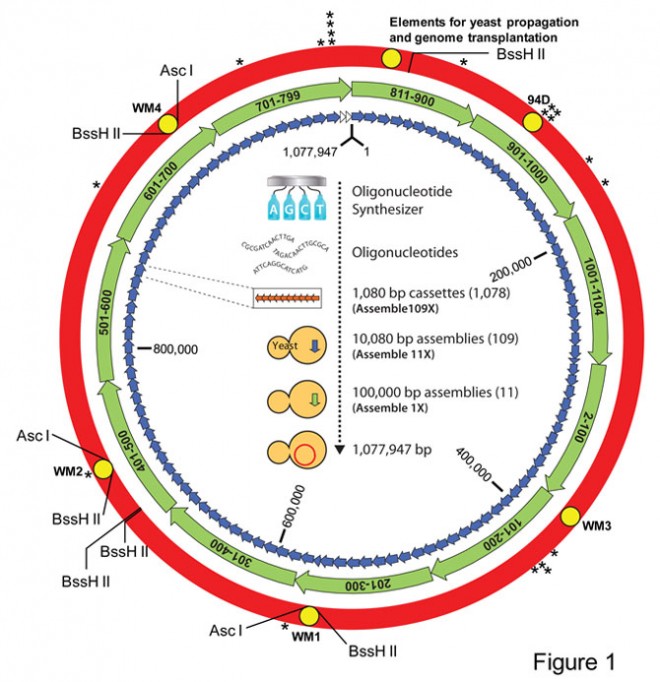
Images: 1) Schematic demonstrating the assembly of a synthetic M. mycoides genome in yeast./Science/AAAS. 2) Images of the phenotype of the JCVI-syn1.0 and WT strains./Science/AAAS.
See Also: